Endothelin-2 Differential Expression in Normal and Early-Stages of Colon Cancer Development ()
1. Introduction
Endothelin family includes three cyclic peptides (ET-1, ET-2 and ET-3) of 21-amino acid with two disulfide bridges established between cysteine residues located at positions 1 - 15 and 3 - 11, and two general types of receptors (ETA and ETB). ET-2 and ET-3 differ from ET-1 in two and six amino acid residues respectively. In mice, a homolog of ET-2 that only diverges in one amino acid residue has been called vasoactive intestinal contractor (VIC) [1]. It is widely accepted that endothelin axis is closely related with several pathologies including cardiovascular disorders, pulmonary disease, renal diseases, cancer and many others [2]. Endothelin axis also takes part in the physiology [3-6] and pathology of gastrointestinal tract [2] although their function is not clear yet.
Alterations in the expression of ET-1 ligand and the receptors ETA and ETB have been shown in human colon cancer. While in premalignant adenomas and malignant colon carcinomas, exhibit increased levels of preproET-1, endothelin-converting enzymes, and ET-1 [7,8], there is no consensus about how the ETA and ETB receptors behave [9,10]. On the other hand, it has been recently proved that ET-2 and ET-3 are silenced in colon cancer and their induced overexpressions inhibit cell migration and invasion [11]. In that work, it was hypothesized that ET-2 and ET-3 might be silenced in early stages of cancer avoiding “mixed messages” in the ET axis. Both the disappearance of ET-2 and ET-3 and the overexpression of ET-1 could lead to the cancer progress. In the present report, we prove that this relation is not linear for ET-2, since its gene expression goes down only in distal colon region.
2. Materials and Methods
2.1. Animal Proceedings
Adult male BALB/c mice (n = 10) 20 - 30 g body weight, was purchased from Center of Biological Experimentations and Biotery of National University of Litoral, Santa Fe. Argentina. Mice were killed by cervical dislocation and colon segments (proximal and distal) were removed. Our experimental procedures were in accordance with the Guidelines on Handling of Laboratory Animals for our institution.
2.2. Murine Model of Chemical-Induced Colon Cancer (AOM/DSS)
Adult male BALB/c mice were intraperitoneal injected with Azoxymethane (AOM)10 mg/Kg body weight and a week later, Dextran Sodium Sulphate (DSS) was given in the drinking water for seven days following the procedure reviewed by Rosenberg et al. (2009) [12], Tanaka (2009) [13] and De Robertis et al., (2011) [14]. Control animal were injected with saline solution and were not given DSS. Samples were taken at day cero and at week four after injection. Proximal and distal segments were destined to histological procedure, immunofluorescence analysis and semi-quantitative RT-PCR for their study.
2.3. Histological Procedure
Colon segments were Carnoy’s fixed (2 h, room temperature) dehydrated in ethanol series and paraffin embedded. Samples were cut at 4µm thick in microtome, dewaxed, rehydrated in a decreasing ethanolic series and Eosin and Hematoxylin stained.
2.4. Immunofluorescence
The immunoreactions were made on 4 μm-thick microtome (Reichert, Austria) sections. The sections were permeated with Triton X-100 0.3% in PBS. To avoid non-specific reactions, sections were blocked with heatinactivated normal goat serum and heat-inactivated normal donkey serum, as required. The antibodies used were rabbit polyclonal IgG (Immuno-Biological Laboratories Co., Ltd., Gunma, Japan) against ET-1, diluted 1:50, goat polyclonal IgG against ET-2 (Santa Cruz, sc-21627), diluted 1:50, rabbit polyclonal IgG against ETA receptor (Santa Cruz, sc-33535), diluted 1:50 and rabbit polyclonal IgG (Immuno-Biological Laboratories Co., Ltd., Gunma, Japan) against ETB receptor, diluted 1:50. The secondary antibodies were goat anti-rabbit IgG-Cy3 (Chemicon, AP187C) and donkey anti-goat IgG-Cy3 (Chemicon, AP180C). Control staining was carried out jumping the primary antibody to the dilution buffer. After reaction, nuclei were stained with Hoechst 33342 (Sigma B-2261). Finally the sections were mounted using Vectashield, fluorescence mounting medium (Vector Laboratories) and observed and recorded using an Olympus BX-50 epifluorescence microscope equipped with an Apogee AM4 CCD camera.
2.5. Semi-Quantitative PCR
Samples were RNA-extracted with TRIzol reagent (Invitrogen) and retro-transcribed with Revert Aid First Strand cDNA Synthesis Kit (Fermentas) using oligo (dT) primers to synthesize cDNA only from mRNA. The cDNAs obtained were PCR amplified and solved in an agarose gel. The primer sequences for each amplified gene are listed in Table 1. The housekeeping gene GAPDH was used as internal control and each analyzed gene was normalized using this control by gel-analysis software, developed by ourselves [15].
2.6. Non-Linear Optical Microscopy
Two-photon excitation fluorescence (TPEF) and second-harmonic generation (SHG) images were acquired with an inverted microscope Axio Observer Z.1 (Carl Zeiss AG, Germany) equipped with a Zeiss LSM 780- NLO confocal scan head (Carl Zeiss AG, Germany), as was recently described in detail [16]. TPEF and SHG images were excited with 20 mW (at the sample) of a 940 nm fs beam, generating a SHG at 470 nm and a TPEF > 490 nm.
2.7. Statistical Analysis
The results are presented as means ± SD and analyzed by unpaired t-test with Welch correction to determine signi- ficant differences among data groups. P values lower than 0.05 were considered statistically significant and lower than 0.01 were very significant.
3. Results
3.1. Cancer Induction Model
We run this experience in order to study the behavior of

Table 1. Optimal primers for real-time PCR of murine endothelin system and GAPDH.
endothelin axis in the initial step of cancer development. At second week of the experience, while animals were drinking DSS, they suffer diarrhea and anal bleeding probably caused by the DSS-generated inflammation. At week three all of them started getting better and they looked completely recovered at fourth week. Comparing with normal colon (Figure 1(A)), Eosin and Hematoxylin histology showed signs of inflammation and aberrant crypt foci and occasionally little microadenomas (Figure 1(B)) as was previously informed by De Robertis et al., (2011), especially in distal colon.
3.2. Immunofluorescence
In normal conditions, ET-2 was mainly immunolocalized at the base of distal colon crypts (Figure 1(C)), in contrast with proximal colon where the localization is much more changeful, including middle and upper part of the crypts. Furthermore the quantification of fluorescence in the immunoreaction gave significant differences between the segments, having proximal colon higher expression than distal one (not showed). Contrary to ET-2, ET-1 was homogeneously expressed both in proximal and distal region. ETA receptor was localized weakly in distal colon, mainly in the basal membrane (Figure 1(E)), while its immunolocalization is significant higher in proximal colon (not showed). On the other hand, ETB receptor was present in the nuclei of epithelial and some muscle cells, according with the observations of Takizawa et al., (2004) [6], Bianchi et al., (2012) [4] and several reports that have proved that the density of ETB receptor is higher in the nucleus than in the cytosol of all human cell types tested [17-20].
We studied distal colon in early stages of cancer induction, where the major rate of tumors is reached in this mice model [12]. The results of this preliminary study have demonstrated that ET-2 peptide was not localized in all the analyzed crypts, but the peptide seems to be reduced in epithelial cells while increase in immune cells like lymphocytes (Figure 1(D)). ET-1 peptide and ETB receptor did not show significant differences in normal and induced animals, ETA receptor seem to reach a higher expression in tissue-infiltrating lymphocytes (Figure 1(F)).
3.3. Gene Expression
In normal mice, the semi-quantitative gene expression analysis showed that the mean of ET-2 transcripts in distal colon was significantly lower than in proximal one (Figure 2(a)), these results seems to be in line with the quantification values of immunofluorescence. Despite having a tendency, the other analyzed genes gave no significant differences (Figure 2(a)). In early stages of induced cancer mice, ET-2 was not detected in distal colon.

Figure 1. Histological sections of E&H-stained slices from normal (A) and AOM/DSS induced animals (B). Immunofluorescence of ET-2 in normal (C) and AOM/DSS induced animals (D), and immunofluorescence of ETA receptor in normal (E) and AOM/DSS induced animals (F), n = 5.
Although the other analyzed genes in the induced model did not exhibit significant differences between proximal and distal colon, they did display the same tendency that was observed in normal mice colon (Figure 2(b)).
There were no significant differences between ET-2 gene expression in ascending colon between normal and pathological mice. By the contrary, in descending colon, the peptide in the induced cancer model diminished its expression level and it could not be detected by the used approach (Figure 2(c)).
Comparing ETA expression in normal and pathological conditions, it was verified that the gene was overexpressed both in proximal and distal colon, being the differences very significant in distal and significant in proximal colon (Figure 2(c)).
3.4. Non-Linear Optical Microscopy
Crypts were identified with TPEF and the connective tis-
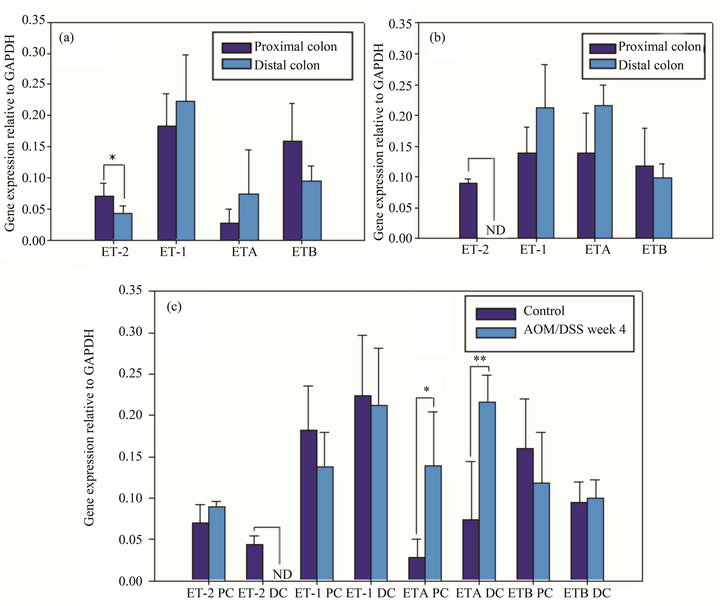
Figure 2. Semi-quantification of gene expression relative to GAPDH of ET-2, ET1, ETA and ETB receptors in proximal and distal portions of control animals (a). Semi-quantification of gene expression relative to GAPDH of ET-2, ET1, ETA and ETB receptors in proximal and distal portions of AOM/DSS induced animals (b). Comparison of the gene expression both in proximal (PC) and distal colon (DC) and in control and AOM/DSS treated animals (c). n=5, ND: not detectable, *(p < 0.05), **(p < 0.001).
sue between crypts was detected via SHG of collagen (Figure 3). In the early stage of tumor tissue, SHG images showed changes in the fibrous stroma. Additionally to collagen fibers morphology, collagen density changes were detected (Figure 3).
4. Discussion
Since most of the current studies are focused on wellestablished colon cancer model behavior, the main objective of the present work was to analyze the endothelin system in early stages of colon cancer development. In normal mice, endothelins are highly expressed in the gastrointestinal tract [3,21,22]. The previously unreporteddifferential distribution of ET-2 (both mRNA and peptide) along colon must be focused in order to understand how this peptide acts. This particular distribution is more interesting yet, when we verify that within the crypt axis this peptide is not uniformly expressed, being mainly immunolocalized at the base of distal colon crypts, whereas in proximal colon it was localized also at the middle and upper part of the crypts. This expression pattern suggests a pleiotropic activity, which seems to be more extent in proximal colon than in distal one.
This particular distribution is deeply modified in early stages of the mice AOM/SDD colon cancer model showing a significant decrease of the mRNA-ET-2 expression on distal colon segment, where it became no detectable. The immunofluorescence pattern is coincident with these results, since most of the crypts are negatives for that peptide and only a weak reaction can be seen. Coincident with our results, recent studies on colon cancer induction models, have reported an epigenetic silencing of the ET-2 gene several weeks before the onset of frank tumors [11]. As was previously mentioned, distal colon exhibit the major tumors rate in early stages of cancer induction [12], therefore ET-2 down regulation

Figure 3. Eosin and hematoxylin, SHG and TPEF images obtained from colon of normal and AOM/DSS week 4 treated mice. Barr: 200 µm.
may be an early prognostic marker of the disease. We hypothesize that at this stage silencing could start in distal portion, and lengthen to the rest of the colon progresssively, while malignancy increases. Although it was reported that significantly elevated ET-1 expression occurs in 80% of primary human colon cancers [23,24], we found no significant differences comparing ET-1 gene expression in normal and early stage colon cancer conditions, thus this peptide might became overexpressed just in later stages.
The ETA receptor plays an important role in tumor cell migration, metastasis, and proliferation. The ETB receptor plays a critical role in angiogenesis and the inhibition of anti-tumor immune cell recruitment [25]. In this work, ETA receptor underwent an overexpression both in proximal and distal segments; this behavior was reported several times in many kinds of tumors [26-32], including colorectal cancer [9]. Recent evidence showed that ETA receptor is expressed on CD133+ cancer stem cells in both cell lines and primary human tumor cells [25] and that CD133 expression exhibit enhanced tumorigenicity over CD133-negative (CD133-) cells, thus the former would be a more malignant phenotype than the latter [33]. Increased expression of ETA receptor in distal colon in early stage of cancer could be related to its increasing presence in cancer stem cells. Furthermore, ETA receptor inhibition prevents the induction of ICAM1, necessary for immune cell recruitment [25]. Since we observed ETA immunoreactivity in lymphocytes, we could hypothesize that its increased gene expression would respond to the increased number of infiltrated lymphocytes. In contrast, ETB receptor, kept on in the same level expression, despite being reported it is down-regulated in colorectal cancer [9] this does not seem to occur in the early stages of the disease.
Collagen forms the structural network of the extracellular matrix (ECM) in tissue and plays an important role during epithelial tumor progression. SHG has already been shown to have potential applicability for cancer diagnosis by revealing changes in the ECM in tumors relative to normal tissues [34]. An advantage of SHG compared with other visualization techniques, such as bright field H&E images, is the SHG selectivity, which is blind to the background populated with too many other things and only sees the collagen fibril’s pattern. This allows an automatic digital imaging processing of collagen network only. The results obtained in this work from SHG are in line with previous studies [35-37]. Jain and coworkers demonstrated that an increase of collagen concentration (i.e., desmoplasia) was associated with implanted melanoma-cell tumor [38]. On the other hand, in a complete analysis of human gastrointestinal tract, Nathanson and coworkers find out small amount of collagen in benign tissues [39]. Furthermore, in a recent study was demonstrated that ET-1 stimulates processes involved in tumor stroma production by colonic fibroblasts [40]. Because the collagen deposition in stroma is a dynamic process which regulates epithelial proliferation/tumor formation cycle, future studies must be performed. However, the present results suggest that collagen concentration could be an important factor in tumoral progression and can be used also as biomarker.
5. Conclusion
Our findings emphasize the idea that the colon must not be considered as a homogeneously functional structure, at least in the endothelin system behavior, according to our study, it allows us dividing it in at least proximal and distal portions. Furthermore, our work found that at early stages of colon cancer, the first modifications in the endothelin axis are the decrease of ET-2 in distal portion and the overexpression of ETA receptor in whole colon.
Since ET-1 acts as a strong stimulus for tumor stroma secretion, collagen generation in the tumor might be increased as a result of ET-1 overexpression. As the disappearance of ET-2 occurs before that, we support that ET-2 decrease might serve as an early biomarker of colon cancer risk, even more than stroma or ET-1 increment.
6. Acknowledgements
This paper was supported by the PID-UNER 6116. The authors are grateful to CEPOF (Optics and Photonics Research Center, FAPESP) and National Institute of Photonics Applied to Cell Biology (INFABIC), UNICAMP, Campinas, Brazil.
NOTES